
Aetos Pharma can provide its clients the customized manufacturing as per the client’s need with their brand name, attractive packaging, and quality material needed keeping in mind the respective regulatory requirements of the country.
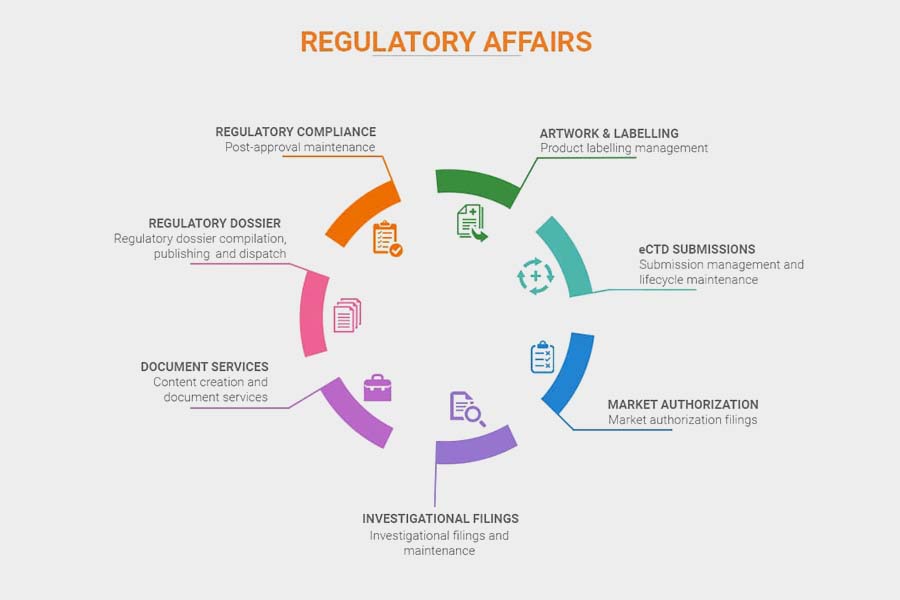
Our team at Aetos Pharma helps our international clients in dossier filling as per their regulatory requirements of respective countries in CTD, ACTD, and eCTD formats.
We help in the complete process starting from dossier preparation, dossier submission, and queries in dossiers from respective ministries of health to our international clients in various countries globally.
We also provide the COA (certificate of analysis), MOA (method of analysis), FSC (free sale certificate), COPP / CPP ( certificate of pharmaceutical product), PP ( product permission), etc all the documents required for the import license/import permit to our clients.

Aetos Pharma can provide technology transfer starting from API sourcing, equipment/machinery procurement, and formulation of finished dosage form from lab scale to commercial development and packaging.
Aetos Pharma Private Limited is a leading WHO-GMP EU-GMP Pharmaceuticals Manufacturer exports products to the following continents
Angola | Benin | Botswana | Burkina Faso | Burundi | Cameroon | Cape Verde | Central African Republic | Chad (Tchad) | Comoros | Republic of the Congo | Democratic Republic of the Congo | Côte d’Ivoire (Ivory Coast) | Djibouti | Equatorial Guinea | Egypt (Misr) | Eritrea | Ethiopia (Abyssinia) | Gabon | The Gambia | Ghana | Guinea | Guinea-Bissau | Kenya | Lesotho | Liberia | Libya | Madagascar | Malawi | Algeria | Mali | Mauritania | Mauritius | Morocco (Al Maghrib) | Mozambique | Namibia | Niger | Nigeria | Rwanda | São Tomé and Príncipe | Senegal | Seychelles | Sierra Leone | Somalia | South Africa | South Sudan | Sudan | Swaziland (Eswatini) | Tanzania | Togo | Tunisia | Uganda | Western Sahara | Zambia | Zimbabwe
Afghanistan | Armenia | Azerbaijan | Bahrain | Bangladesh | Bhutan | Brunei | Cambodia (Kampuchea) | Georgia | India | Indonesia | Iran | Iraq | Japan | Jordan (Al Urdun) | Kazakhstan | Kuwait | Kyrgyzstan | Laos | Lebanon (Lubnan) | Malaysia | Maldives | Mongolia | Myanmar (Burma) | Nepal | North Korea | Oman | Philippines | Qatar | Russia | Saudi Arabia | Singapore | South Korea | Sri Lanka | Syria | Tajikistan | Thailand | Turkey | Turkmenistan| Taiwan | United Arab Emirates | Uzbekistan | Vietnam | Yemen
Albania | Andorra | Austria | Belarus | Belgium | Bosnia and Herzegovina | Bulgaria | Croatia | Cyprus | Czech Republic | Denmark | Estonia | Finland | Georgia | Germany | Greece | Hungary | Iceland | Republic of Ireland | Italy | Kosovo | Latvia | Liechtenstein | Lithuania | Luxembourg | North Macedonia | Malta | Moldova | Monaco | Montenegro | Netherlands | Norway | Poland | Portugal | Romania | San Marino | Serbia | Slovakia | Slovenia | France | Spain | Sweden | Switzerland | Ukraine | United Kingdom
Canada | United States of America | Navassa Island | Puerto Rico | US Virgin Islands | Guam | American Samoa
Brazil | Argentina | Bolivia | Chile | Colombia | Ecuador | Falkland Islands | French Guiana | Guyana | Paraguay | Peru | South Georgia and the South Sandwich Islands | Suriname | Uruguay | Venezuela | Belize | Costa Rica | El Salvador | Guatemala | Honduras | Nicaragua | Panama | Haiti | Antigua and Barbuda | Bahamas | Barbados | Cuba | Dominica | Grenada | Jamaica | Saint Lucia | Saint Kitts and Nevis | Saint Vincent and the Grenadines | Trinidad and Tobago
Australia | Fiji | New Zealand | Federated States of Micronesia | Kiribati | Marshall Islands | Nauru | Palau | Papua New Guinea | Samoa | Solomon Islands | Tonga | Tuvalu | Vanuatu
